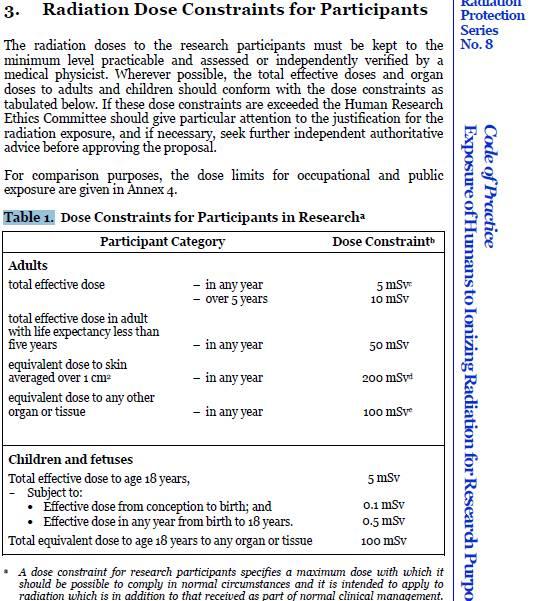
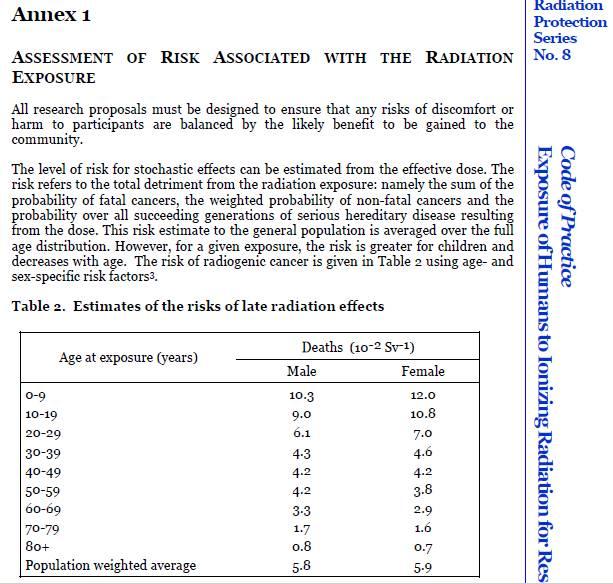
Radiation ExposureThe Code of Practice – Exposure of Humans to Ionizing Radiation for Research Purposes outlines the requirements a researcher must take to ensure participants are notified of additional radiation exposure. The code states: The Code of Practice requires researchers to have dose calculations and associated risk information provided to participants and HREC’s checked by an independent medical physicists in the relevant field. In the case of doses in excess of certain dose constraints, verification by a second medical physicist must be obtained. The researcher must provide the research participant with sufficient written information about the purpose, methods, radiation dose, associated risks and any discomforts of the radiation exposure to enable the research participant to give informed consent. Article 2.1.6 (page 4) The researcher must obtain an independent assessment of verification by a medical physicist of: (a) the total effective dose 1 and relevant organ doses for those radiological procedures that are performed specifically for the research protocol and which are additional to those received as a part of the research participant’s normal clinical management; (b) whether these will exceed the dose constraints in Table 1 (page 15); and (c) the risks associated with the radiation exposure in accordance with Annex 1 (page 21)
Office Requirements For NSW/QLD only – a letter from the PI stating that radiation exposure is part of routine care OR If radiation exposure is additional to that received an independent assessment report by a Medical Physicist. For VIC – complete section 4 of the Victorian Specific Module for each site For Western Australia – complete the Western Australia Specific Module for each site
|