Meeting DatesLow or Negligible Risk ApplicationsThere are no closing dates for submitting Low Risk HREA projects. Applications can be submitted to the Office at anytime and are reviewed out of session by the HREC on an ad hoc basis.
Greater than Low Risk ApplicationsGreater than low risk applications are processed and then assigned to a full HREC meeting. Please note the closing dates for submission and related meeting dates below:
GREATER THAN LOW RISK HREA - MEETING DATES FOR 2025/2026 Note: The November 2025 meeting date has been amended on 28/10/2025. The new date is shown above in the table. Please note that you will receive feedback on the initial review of your Greater than Low Risk Application within 1 week after the HREC meeting date.
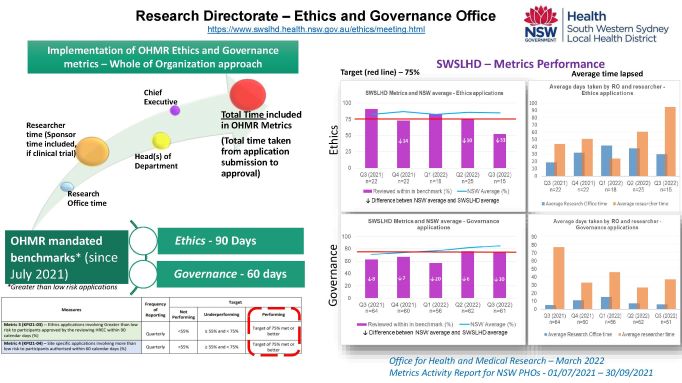
The Office of Health and Medical Research (OHMR) has amended the Key Performance Indicators (KPIs) for Ethics and Governance applications. Since July 2021, the new KPIs include total time taken by all stakeholders, including research office, Head of Departments, researchers/sponsors etc. Please note that the mandated timeframes for processing are;
|